 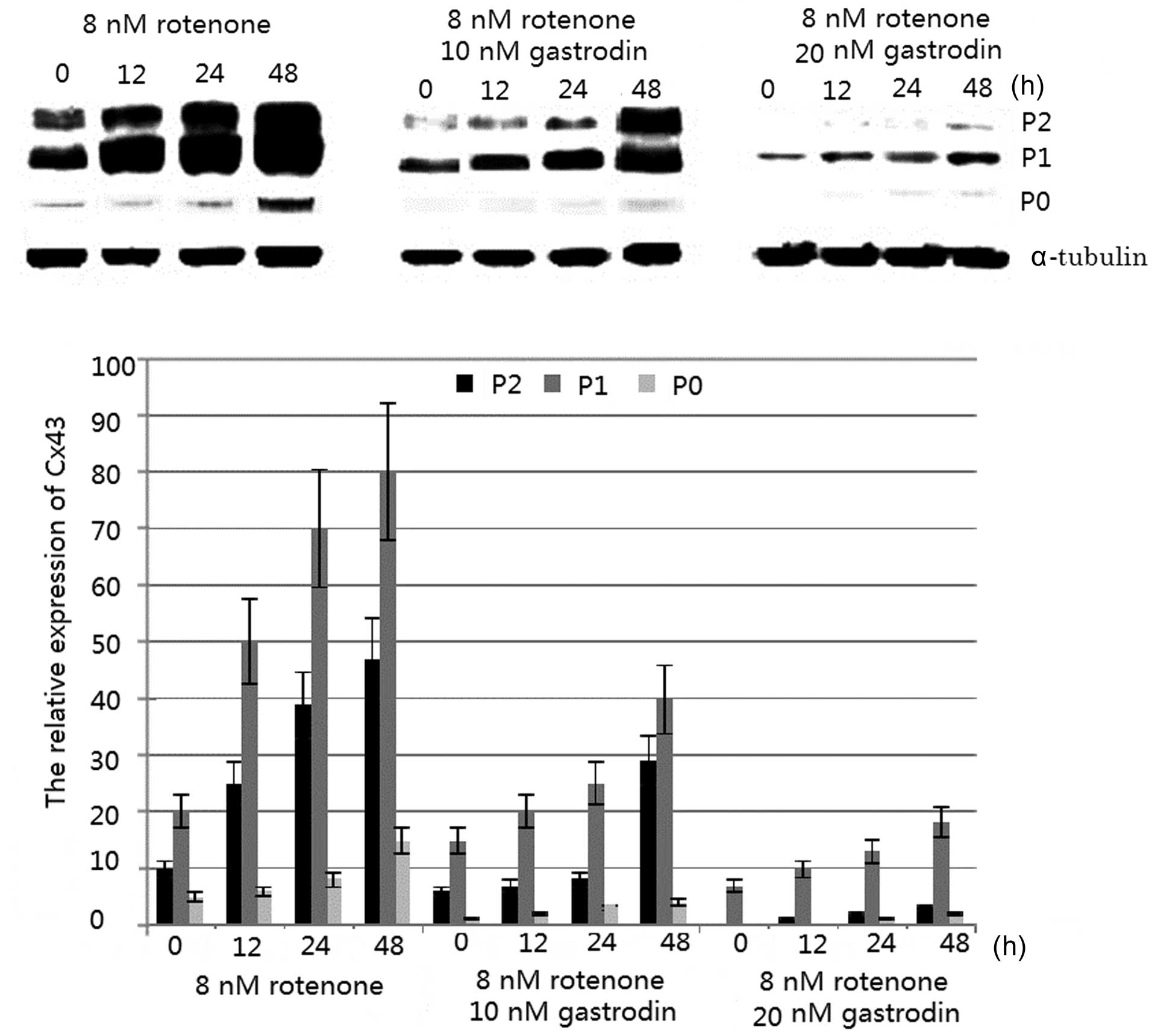 
Normalize: An accurate normalization strategy can correct for minor variation that occurs in spite of careful experimental technique. To ensure each sample begins with the same amount of protein, a protein concentration assay must be used to adjust sample concentration and make sample loading across your gel as consistent as possible. Examples in this section will show why the following points are important to consider when you plan an experiment.Įnsure even sample loading: Differences between protein abundance on a blot cannot be determined unless the same amount of sample protein is loaded in each lane. However, a well-designed protocol will reduce the impact of this inherent variation on your results. Small sample-to-sample and lane-to-lane variations are inevitable. This guide focuses on examples of how normalization can go wrong, and provides tips for avoiding such problems. The Normalization Handbook ( /handbook) describes how to choose and validate an appropriate internal loading control. The ILC must not interfere with target detection.Ĭhoosing and validating an ILC that meets these requirements is fundamental to the design and accuracy of a quantitative Western blot. The ILC and target must be detected within the same linear range. The ILC must be unaffected by experimental conditions. To ensure that your strategy is accurate, verify that your ILC meets these requirements: Normalization Requirementsįor a normalization strategy to be accurate, it must conform to the core principle of normalization. Target and internal loading control signals must vary to the same degree with sample loading (11). Linear range (LR): The span of signal intensities that display a linear relationship between amount of protein on the membrane and signal intensity recorded by the detector.Īccurate normalization requires that the target and internal loading control signals are both entirely dependent on sample loading. Total protein staining of the membrane after transfer to visualize actual sample loading in each lane.An unrelated internal reference protein, typically a housekeeping protein (HKP, such as actin, tubulin, or GAPDH) that is expressed in all samples at a stable, constant level.Several types of ILC can be used, including: Internal loading control (ILC): Endogenous protein(s) that are unaffected by experimental conditions and used as an indicator of sample loading (11). The target and internal loading control must be detected within the same linear range. Normalization mathematically corrects for unavoidable sample-to-sample and lane-to-lane variation by comparing the target protein to an internal loading control. Inaccurate normalization has many causes, including: This guide will present examples where normalization has been adversely affected by common pitfalls and will offer potential solutions. Irregularities across the blot due to problems such as inconsistent transfer, uneven blocking, or uneven antibody incubation However, when used properly, normalization can minimize many common sources of variation, such as: No single normalization method can correct for everything. Several normalization methods are available, but the goal of each is to correct for variability inherent to Western blotting. In simple terms, normalization is a way to improve the quantitative accuracy of Western blot data by correcting for small lane-to-lane and sample-to-sample variation. Figure 2 shows a stylised western blot of increasing concentrations of protein, and the “signal intensity” as measured by a commonly used software-in this example the last five concentrations gave the same intensity measurement despite representing very different amounts of protein.Good Normalizations Gone Bad Introduction This represents a general problem of quantifying western blots with simple image analysis software, which may be unable to discriminate between similar-looking bands that have fallen off the end of the linear scale. The chemiluminescent film was saturated, so the higher level of tubulin in the wild type was not reflected when the intensity measurements were taken: actually when the same amounts of sample were loaded, there was no change in expression of Protein X in the two conditions. In fact, the gel for the wild type was accidentally loaded with more of the sample. However, although the two tubulin controls look the same-and give the same intensity measurements using a simple image analysis tool-they do not represent the same underlying expression. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
